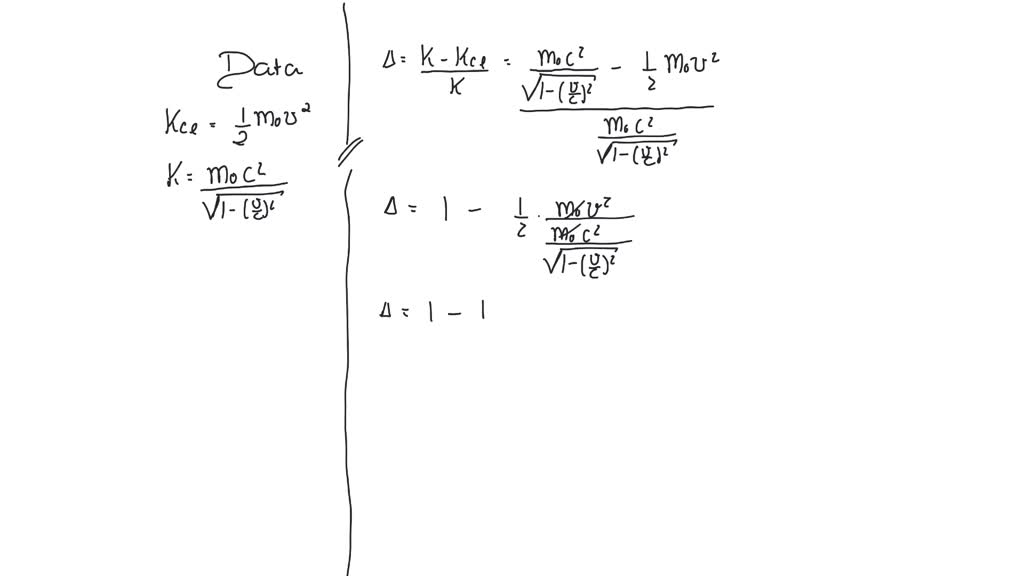
SOLVED: What is the percent difference between the classical kinetic energy, Kcl=12m0v2, and the correct relativistic kinetic energy, K=m0c2/1−v2/c2−−−−−−−√−m0c2, at a speed of 0.11 c? Express your answer using two significant figures.

Amazon.com: BulkSupplements.com Potassium Chloride Powder - Potassium Supplement for Muscle Support & Electrolytes - Gluten Free, Soy Free, and No Filler Powder, Potassium Salt (500 Grams - 1.1 lbs) : Health & Household

SOLVED: (E1) (E2) (E3) (E4) Fi2 k"r12 E=kCg;; 5 = f E"dA Lnc E = k f %; (C1) C2 C3 C4) C5 C6) (E5) (EG) (E7) Vi = k94 V=kCL V =
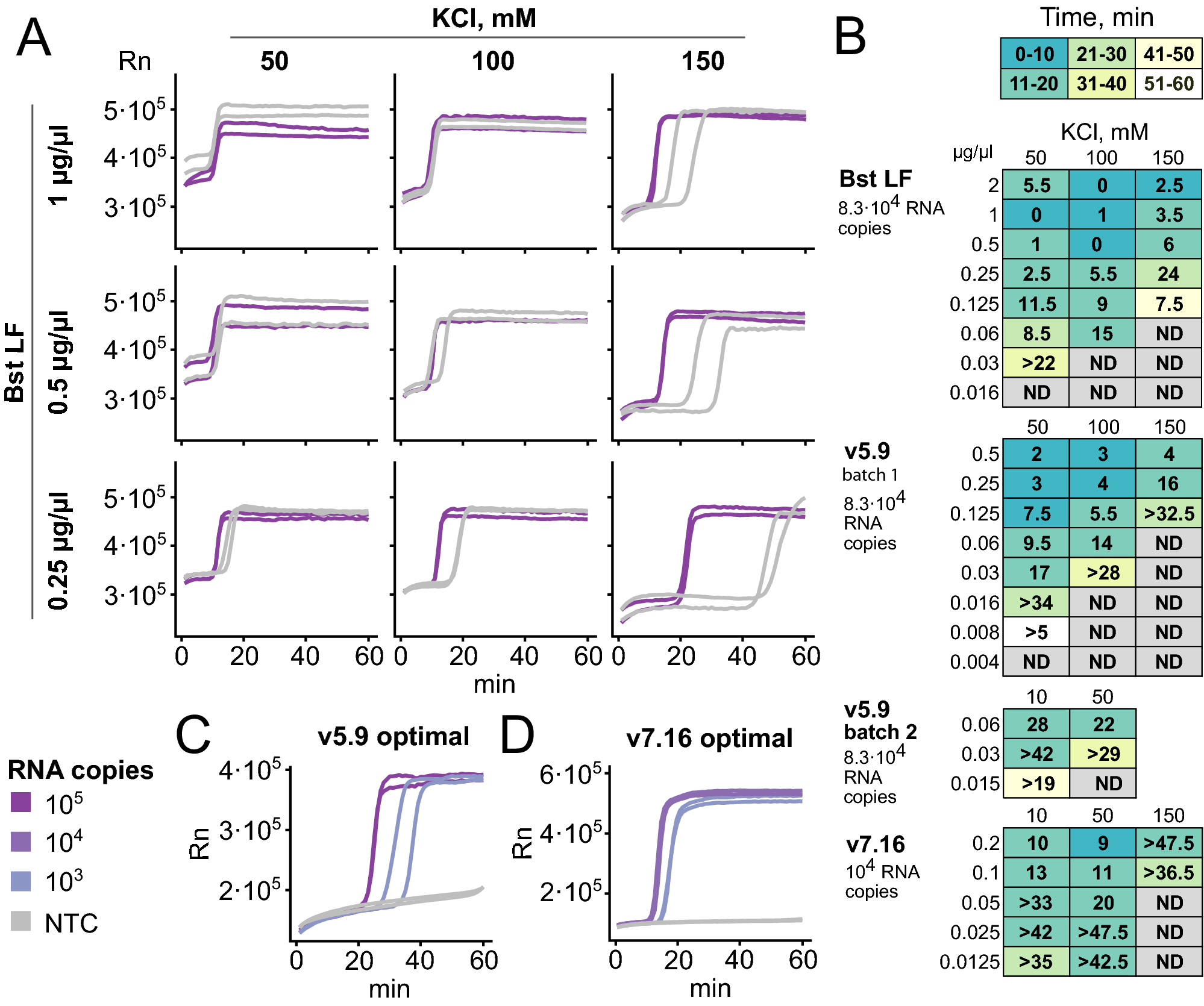
Direct detection of SARS-CoV-2 using non-commercial RT-LAMP reagents on heat-inactivated samples | Scientific Reports
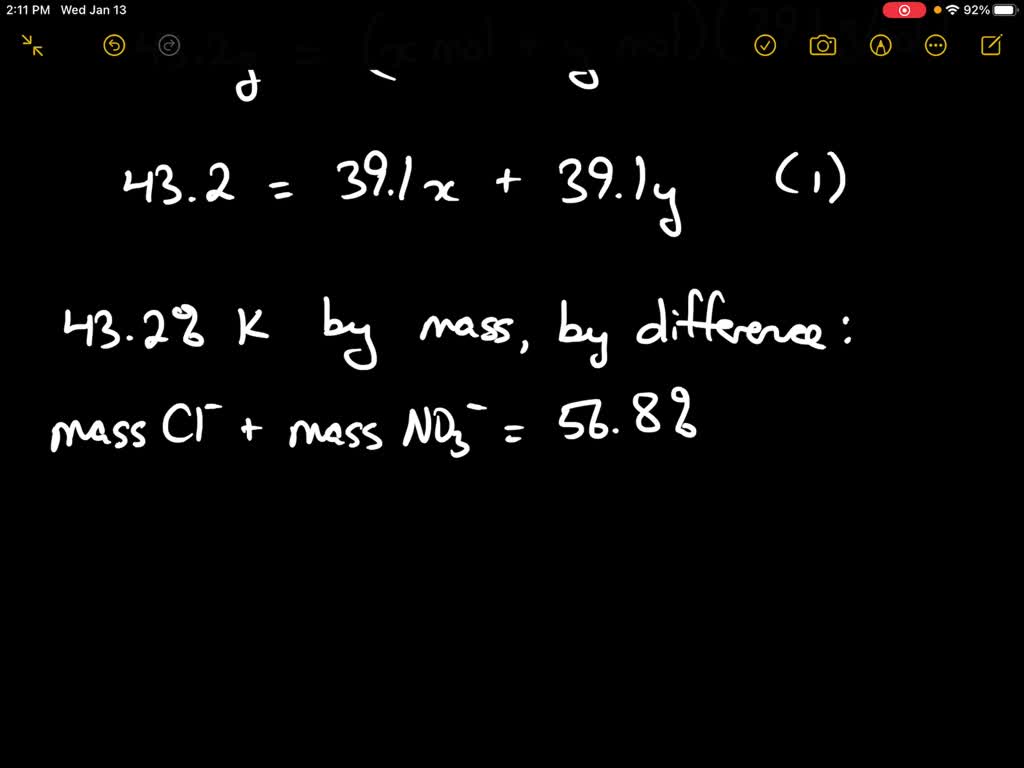
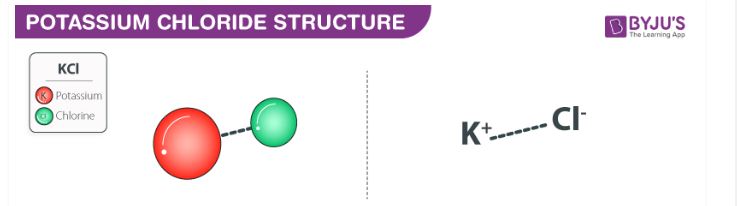
SOLVED: El peso molecular del KCl (Cloruro de potasio)es: * 74,54 g/mol 35,45 g/mol 73, 54 g/mol 46,45 g/mol

Distinction between Human Cytochrome P450 (CYP) Isoforms and Identification of New Phosphorylation Sites by Mass Spectrometry | Journal of Proteome Research

NaCl and KCl mediate log increase in AAV vector particles and infectious titers in a specific/timely manner with the HSV platform - ScienceDirect

SOLVED: Use the following information to calculate the heat of sublimation for potassium (K): Heat of formation for KCl(s) = -437 kJ/mol Electron affinity for Cl = -349 kJ/mol Ionization energy for

A particularly simple NH4Cl-based method for the dissolution of UO2 and rare earth oxides in LiCl-KCl melt under air atmosphere - ScienceDirect